Dr. Dominick J. Casadonte, Jr.

Title: Minnie Stevens Piper Professor
Education: Ph.D., Purdue University, 1985
Postdoctoral Study, University of Illinois, 1985-89
Dreyfus Foundation Scholar/Fellow, 1988-89
Fulbright Senior Scholar (France), 2000
NSF Discovery Corps Fellow, 2004-05
Fellow, American Chemical Society, 2019
President's Award for Excellence in Science, Mathematics, and Engineering Mentoring
(PAESMEM), The White House, 2019
Research Area: Inorganic & Materials Chemistry
Office: Chemistry 226-A
Phone: 806-834-2746
Fax: 806-742-1289
Email: dominick.casadonte@ttu.edu
Webpage: Research Group
Principal Research Interests
- The Chemical Effects of High-Intensity Ultrasound
- Nanomaterial Fabrication
- Environmental Remediation
- Catalysis
- Supramolecular Chemistry
- Photophysics/Photochemistry
- Chemical Education
Professor Casadonte has three areas of active research: 1) The application of high-
and low-intensity ultrasound in the fabrication of novel nanomaterial and molecular
systems with potential medical, energy storage, catalytic, and environmental remediation
activity, 2) The synthesis of supramolecular first- and second-row transition metal
organometallic complexes for use as molecular photo-circuit elements. Many of these
compounds possess unusual excited state processes which we strive to understand, and
3) Chemical education Professor Casadonte has a variety of interests here. As a leader
in the flipped classroom movement, Professor Casadonte performs research on this methodology
for effective classroom utilization. He is also known for his work in chemical laboratory
safety education.
The Chemical Effects of High-Intensity Ultrasound
Acoustic cavitation produces temperatures in excess of 5000 K and pressures greater
than 100 atmospheres during the adiabatic collapse of gas vacuoles in solution. These
physical extremes are used to produce high-energy species not available by analogous
thermal or photochemical routes, and the shockwaves and radiation pressure concomitantly
produced can generate unique materials formation. Sonication of mixed-metal powders
in hydrocarbon solvents leads to the formation of intermetallic coatings which may
have use as dehydrogenation catalysts and in thin film coatings. Some of our other
activities involving sonochemistry in materials synthesis include the fabrication
of metal phosphide semiconductor materials from organometallic precursors, generation
of graphite intercalation compounds, facile sonochemical methodologies for the formation
of imidazolium- and thiophene-based ionic liquids, and the formation of true nano-alloys
of precise stoichiometry for fuel cell catalysis as well as the synthesis of superparamagnetic
materials for medical applications.
The Casadonte group also has an interest in environmental remediation. Ultrasound
is an effective means of removing hydrocarbon contaminants from aqueous media. We
have, for example, been able to oxidatively degrade up to 1500 ppm of a variety of
aliphatic, aromatic, and halogenated hydrocarbons in water in less than one hour.
During his Fulbright Fellowship in France, Dr. Casadonte began preliminary studies
on the application of pulsed ultrasound to enhance sonochemistry. It was found that
with the appropriate pulsed waveform an enhancement of more than 300% in the rate
of degradation of environmental contaminants could be achieved relative to continuous
ultrasonic irradiation.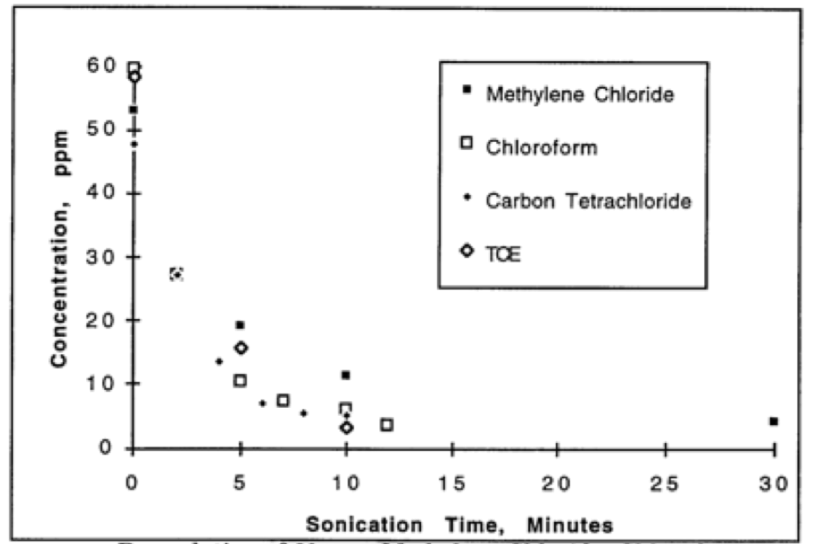
Figure 1. Sonochemical degradation of chlorinated hydrocarbons in water.
Our group is also interested in exploring non-linear cavitation processes using variable
frequency and heterodyne ultrasound. In this case, two different ultrasonic frequencies
are input which produce a third frequency which is non-resonant with the cavitation
sites. It is our belief that the non-linear cavitation phenomena produced by the multi-frequency
ultrasound will lead to enhancements in the rate and efficiency of cavitation. We
have already observed a 500% increase in the rate of acid orange degradation through
the application of heterodyne ultrasound using a multi-frequency sonicator built in
our lab.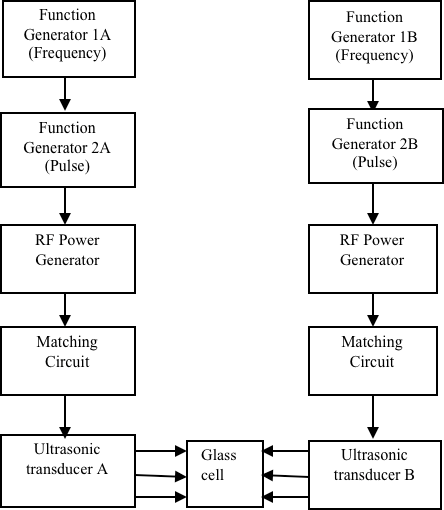
Figure 2. Block Diagram of the Variable Frequency Heterodyne Sonication System (ref.
1)
Supramolecular Chemistry and the Design of Photoactive Metallopolymers
The goal of this research is an understanding of the spatial characteristics required
for the fabrication of molecular photodevices. We have recently developed the first
examples of photoactive multinuclear Cu(I) complexes containing bridging phosphines.
We have also prepared multinuclear Cu(I) complexes containing phenanthrolines bound
via oxygen bridges.![Crystal structure of [(PPh3)2Cu(dmp)-O-(dmp)Cu(PPh3)2]2+ (dmp = 2,9-dimethyl-1,10-phenanthroline)](/chemistry/Faculty/casadonte/molecule1.jpg)
Figure 3. Crystal structure of [(PPh3)2Cu(dmp)-O-(dmp)Cu(PPh3)2]2+ (dmp = 2,9-dimethyl-1,10-phenanthroline)
Similar complexes have been catenated to form metallo-polymers possessing photo-induced
charge-storage capacity. Our research group has recently synthesized a unique series
of Cu(I) metallopolymers containing phenylethynyl and ethynyl linkages between the
phenanthroline units to form a fully conjugated "πway" for electrons to travel after
MLCT photoecitation. Metallopolymers of this type may have future applications in
optical computing.
![Poly-[4,7-diethnyl-1,10-phenanthroline copper(I) bis(triphenylphosphine) tetrafluoroborate]](/chemistry/tetrafluoroborate.png)
Figure 4. Poly-[4,7-diethnyl-1,10-phenanthroline copper(I) bis(triphenylphosphine) tetrafluoroborate] (29)
Photochemistry/Photophysics
Another aspect of Professor Casadonte's research includes the synthesis of complexes
which display simultaneous emission from two or more distinct excited states. He has
recently prepared a series of Cu(I) complexes containing phosphine sulfide ligands
which emit from both charge transfer and intraligand emission excited states. Density
functional calculations are currently underway in an attempt to understand the mechanism
of multistate luminescence.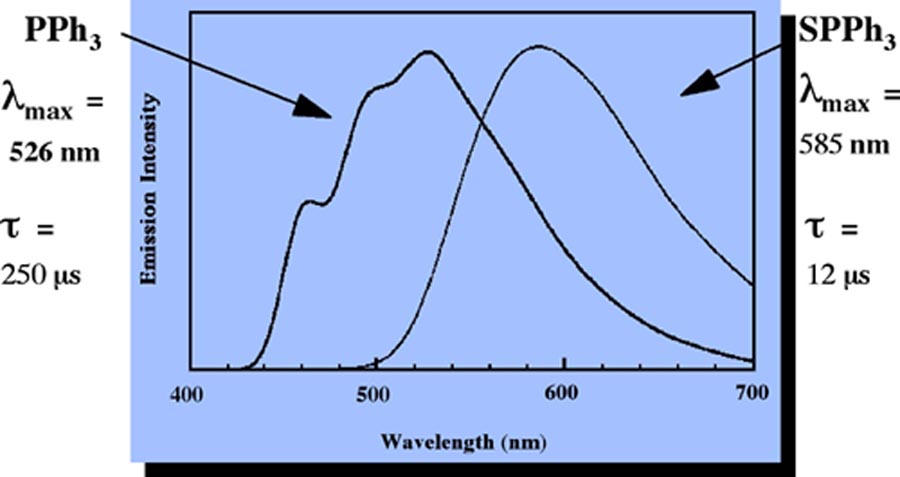
Figure 4. Luminescence Spectra of Cu(I) Phosphine and Phosphine Sulfide Complexes
Chemical Education
Professor Casadonte has an active interest in several areas of chemical education.
In his recent role as a National Science Foundation Discovery Corps Fellow, professor
Casadonte has explored the efficacy of an intergenerational learning and service program
in the chemical sciences in elementary and middle schools, involving the use of senior
citizens as secondary teachers at the 5th and 8th grade levels. Studies were performed
on student learning enhancement, improvement in cognitive functioning among the seniors,
and changes in attitudes between both the seniors and children during their curricular
and extracurricular interactions.
Professor Casadonte has an active program in service learning. He has developed a
laboratory course that teaches chemistry though the development of novel chemical
demonstrations. The students are then required to "perform" their demo show for various
K-12 audiences. Pre- and post-content and attitudinal assessment instruments have
been developed to determine the effectiveness of this hands-on, project-based learning
environment. He is currently working on the development of an intercollegiate service
learning project.
Professor Casadonte was one of the first university chemistry faculty in the country
to develop the concept of "course flipping". In 2017 he was designated by the Flipped
Learning Global Initiative as one of the "Top 40 Flipped Learning Experts in the World".
His recent research in the area of the flipped classroom includes the efficacy of
the methodology, optimum pre-class lecture length, and the effect of peer learning
assistants on the improvement in in-class experience.
Representative Publications
- (Invited Paper) "Ten Years after the Texas Tech Accident. Part II: Changing Safety Cultures and the Current State of Academic Laboratory Safety at Texas Tech University," Eighmy, T., Schovanec, L. Mulcahy, M.B., Young, A., Pappas, D., Martin, J., Coats, H., Franco, J., Casadonte, D.J. ACS Chem. Health Saf. 2020, 27, 3 https://dx.doi.org/10.1021/acs.chas.0c00047
- (Invited Paper) "Ten Years After the Texas Tech Accident: Part I: A Historical Retrospective," Eighmy, T., Schovanec, L., Young A., Martin, J. Casadonte, D.J. ACS Chem. Health Saf. 2020, 27, 2, 105-113
- "Synthesis, Structural Characterization, Photophysical Properties, and Catalytic Evaluations of Transition Metal Complexes of 2,9-Dimesityl-1,10-phenanthroline (dmesp) Ligand," Cetin, M., Shafiei, S., Chen, J. Miller, A., Unruh, D., Casadonte, D., Lohr, T., Marks, T., Stoddart, F., Mayer, M., Findlater, M. J Polym Sci. 2020, 58:1130–1143 DOI: 10.1002/pol.20190276
- "Heterodyne I: Enhancing Sonochemical Efficiency Through Application of the Heterodyne Effect: An Initial Study," Reno, J., Korremula, B., Casadonte, D.J. Ultrasonic Sonochemistry, 2019, 56, 143-149
- "Nanoassembly of perovskite-based photocatalysts in a nanoconfined system for photocatalytic H2 production under visible light," Moniruddina, Md., Meuslinga, B., Duprea, R., Casadonte, D.J. Nurajea, N. Mol. Catal., 2019, 478, 110719-110726, https://doi.org/10.1016/j.mcat.2019.110719
- "Mechanical, rheological, and stability performance of simulated in-situ cured oil well cement slurries reinforced with alumina nanofibers," McElroy, P., Emadi, H., Surowiec, K., Casadonte, D.J., J. Pet. Sci. Eng., 2019, 183, https://doi.org/10.1016/j.petrol.2019.106415
- "The Effectiveness of Course Flipping in General Chemistry – Does It Work?" Casadonte, D.J. ACS Symposium Series "The Flipped Classroom Volume 2: Results From Practice", 2017, 19-37
- "Forensic Identification of Pharmaceuticals via Portable X-Ray Fluorescence and Diffuse Reflectance Spectroscopy," Shutic, S., Chakraborty, S., Li, B., Weindorf, D.C., Sperry, K, and Casadonte, D. Forensic Science International, 2017, 279, 22-32
- "Characterization and Photocatalytic Behavior of 2,9-di(aryl)-1, 10-Phenanhtroline Copper (I) Complexes," Cetin, M.M., Hodson, R.T., Hart, C.R. Cordes, D.B., Findlater, M., Casadonte Jr., D.J., Cozzolino, A.F., Mayer, M.F., Dalton Transactions, 2017, 2017,46, 6553-6569
- "Children's Attitudes and Classroom Interaction In an Intergenerational Education Program." Chorn Dunham, C.; Casadonte, Jr. D.J. Educational Gerontology 2009, 35(5), 453 – 464.
- "Structural Analysis of Sonochemically Prepared PtRu Versus Johnson Matthey PtRu in Operating Direct Methanol Fuel Cells." Stoupin, S.; Rivera H.; Li Z.; Segre C.; Korzeniewski, C.; Casadonte, Jr, D.J.; Inoued, H.; Smotkin, E.S. Phys. Chem. Chem. Phys. 2008, 10, 6430–6437.
- "Applications of Sonochemistry and Microwaves in Organommetallic Chemistry." Casadonte, D.J. Jr.; Li, Z.; Mingos D.M.P. in Comprehensive Organometallic Chemistry III Vol. 1; Edited by Robert H. Crabtree and D. Michael P. Mingos, Elsevier: Oxford, 2007, pgs. 307-340 ISBN: 008044590.
- "Facile Sonochemical Synthesis of Nanosized InP and GaP." Casadonte, D.J. Jr.; Li, Z.; Ultrasonics Sonochemistry 2007, 14(6), 757-760.
- "PtRu Nanoparticle Electrocatalyst With Bulk Alloy Properties Prepared Through A Sonochemical Method." Basnayake, R.; Li, Z.; Katar, S.; Zhou, W.; Rivera, H.; Smotkin, E.S.; Casadonte, D.J. Jr.; Korzeniewski, C; Langmuir 2006, 22, 10446-10450.
- "Hydration and Interfacial Water in Nafion Membrane Probed by Transmission Infrared Spectrscopy." Basnayake, R.; Peterson. G.R.; Casadonte, D.J. Jr.; Korzeniewski, C.; J. Phys. Chem. B 2006, 110, 23938-23943.
- "The Use of Pulsed Ultrasound Technology to Improve Environmental Remediation: A Comparative Study." Casadonte, Jr., D.J.; Petrier, C.; Flores, M. Environ. Tech. 2005, 26(12), 1411- 1416.
- "Enhancing Sonochemical Degradation of Environmental Contaminants Using Power-Modulated Pulsed Ultrasound: An Initial Study.“, Casadonte, Jr., D.J.; Petrier, C.; Flores, M. Ultrasonics Sonochemistry 2005, 12(3), 147-152.
- "Facile Sonochemical Synthesis of Graphite Intercalation Compounds.“, Jones, J. E.; Cheshire, M. C.; Casadonte, Jr., D.J.; Phifer, C.C. Org. Letts. 2004, 6(12), 1915-1917.
- "Photobleaching Comparison of Poly(methylphenylsilylene) and Poly(phenylsilyne)." Thomes Jr., W.J.; Simmons-Potter, K.; Phifer, C.C.; Potter Jr., B. G.; Jamison, G.M.; Jones, J.E.; Casadonte, Jr., D.J. J. App. Phys. 2004, 96(11), 6313-6318.
- "Methanol Electrochemical Oxidation at Nanometer-Scale PtRu Materials.“, Korzeniewski, C.; Basnayake, R.; Vijayaraghavan, G.; Li, Z.; Shanhon, X.; Casadonte, D.J. Surface Sci. 2004, 573 (1), 100-108.
- "The Use of Pulsed Ultrasound Technology to Improve Environmental Remediation: A Comparative Study." Casadonte, Jr., D.J.; Flores, M.; Petrier, C. Environmental Technology 2004, Accepted for Publication.
Department of Chemistry & Biochemistry
-
Address
1204 Boston Avenue, Lubbock, TX 79409-1061 -
Phone
806.742.3067
