Dr. Micah Green's collaborative research paves the way for the synthesis of nanotube fibers
Edited by Javad Hashemi and Jeff Sammons

Dr. Micah Green, a new assistant professor in the Department of Chemical Engineering, has recently co-authored an article available online in the journal Nature Nanotechnology. The research focused on the development of processes to completely disperse carbon nanotubes. Green was a key researcher on the project during his time as a postdoctoral fellow at Rice University in Professor Matteo Pasquali's research group.
Few technological breakthroughs have received as much hype as carbon nanotubes. Since their discovery in 1991, nanotubes have been touted as everything from a cure for cancer to a solution for the world's energy crisis — largely due to nanotubes' extraordinary properties. The hype is all the more remarkable given that nanotubes are notoriously difficult to work with, and that chemists worldwide struggled for years just to make them.
New research results have demonstrated a method for the industrial-scale processing of pure carbon-nanotube fibers that could lead to revolutionary advances in materials science, power distribution, and nanoelectronics. The result of a nine-year program, the method builds upon tried-and-true processes that the chemical industry has used for decades to produce fibers from rigid polymers. The article is available online in the journal Nature Nanotechnology.

Following a 2003 breakthrough with acid solvents, the team methodically studied how nanotubes behaved in different types and concentrations of acids. By comparing and contrasting the behavior of nanotubes in acids with the literature on polymers and rodlike colloids, the team developed both the theoretical and practical tools that chemical firms will need to process nanotube-based materials in bulk through the use of chlorosulfonic acid.
Green played a key role in the computational modeling of nanotube dissolution and liquid crystal formation. The team placed a high priority on understanding the physical forces responsible for the experimental results. Green's work in modeling demonstrated that the experimental methodology could produce a true solution.
Green's modeling demonstrated that chlorosulfonic acid is an athermal solvent, breaking apart the nanotubes from bundles so effectively that they are dissolved as individual tubes without any bundling.
After modeling the interactions, Green worked with Ishi Talmon and his colleagues at the Technion in Haifa, Israel to experimentally prove the breakthrough by inserting the nanotubes into chlorosulfonic acid and freezing the solution. The team used cryo-transmission electron microscopy to analyze the solution and verified that the nanotubes were dispersed as individuals.
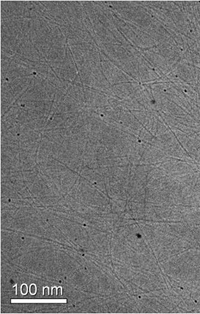
“We took great care to understand what was going on at the molecular level between the individual nanotubes and what was going on at the macroscopic level in the processing of the fiber," Green said. “The modeling and experimentation ensured that we understood why our results are valid, reliable, and replicable for industry.”
The article, “True solutions of single-walled carbon nanotubes for assembly into macroscopic materials”, features the work of an 18-member team of scientists from Rice's Richard E. Smalley Institute for Nanoscale Science and Technology, the University of Pennsylvania and the Technion-Israel Institute of Technology. The effort was led by Pasquali and the lead author is former Rice student Virginia Davis, now a professor at Auburn University.
Green joined the Department of Chemical Engineering in the Whitacre College of Engineering as an assistant professor in August of 2009. A 2002 graduate of Texas Tech University with a Bachelor of Science in Chemical Engineering, Green went on to the Massachusetts Institute of Technology and received a Doctor of Philosophy in Chemical Engineering. After graduation, he worked as a J. Evans Attwell-Welch Postdoctoral Fellow at the Rice University Department of Chemical and Biomolecular Engineering.
To get more information, read the Nature Nanotechnology article "True solutions of single-walled carbon nanotubes for assembly into macroscopic materials".
Edward E. Whitacre Jr. College of Engineering
-
Address
100 Engineering Center Box 43103 Lubbock, Texas 79409-3103 -
Phone
806.742.3451 -
Email
webmaster.coe@ttu.edu
