HISPID COTTON RAT
Sigmodon hispidus Say and Ord 1825
Order Rodentia : Family Cricetidae
DESCRIPTION. A moderately large, robust rat with pattern of last two lower molars S-shaped; tail shorter than head and body, sparsely haired, the annulations and scales clearly visible; ears relatively small and blackish or grayish; pelage coarse and grizzled, the black guard hairs rather stiff (hispid); hind foot with six plantar tubercles, three middle toes longer than outer two; upperparts grizzled brown; underparts grayish white or buff. Dental formula: I 1/1, C 0/0, Pm 0/0, M 3/3 × 2 = 16. Averages for external measurements: total length, 270 mm; tail, 110 mm; hind foot, 31 mm. Weight, 80–150 g.

DISTRIBUTION. Statewide.
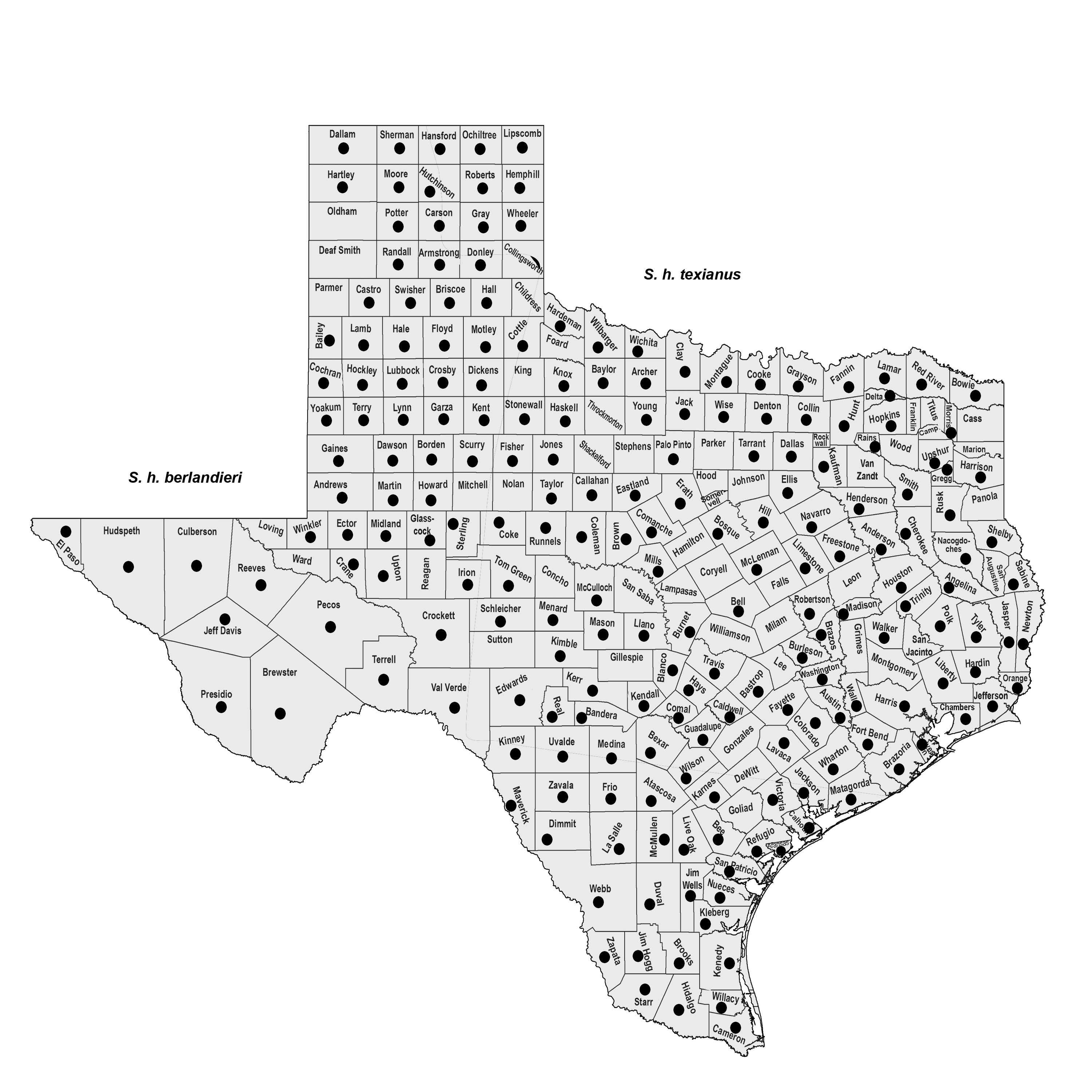
SUBSPECIES. Sigmodon h. berlandieri from the Panhandle southward to the Trans-Pecos and the Rio Grande Plains and S. h. texianus in the eastern and central parts of the state.
HABITS. Normally this cotton rat inhabits tall-grass areas where such grasses as bluestem (Andropogon), cordgrass (Spartina), or sedges (Carex) offer both freedom of movement under a protective canopy and an adequate food supply. In such situations their runways form a network of interconnecting travel ways about 5–8 cm wide. In western Texas, where grassy ground cover is not available, the rats live in dens at the bases of small, low clumps of mesquite in otherwise nearly barren terrain, much after the fashion of white-toothed woodrats. Between those two extremes are several types of habitat that may support small populations of hispid cotton rats. Preferred sites are old fields, natural prairie, unmolested right-of-ways for roads and railroads, grassy and weedy valleys, and dense riparian woodland with thick grass.
The rats place their nests either in chambers off underground burrows or aboveground in dense clumps of grass, piles of brush, or other situations that offer some concealment and protection. The nests are globular, about 12 cm in diameter, and composed of shredded grasses and weeds. Underground burrows are 3–5 cm in diameter, simple in design, and seldom longer than 8 m. Occasionally, the rats take over and use the discarded burrows of pocket gophers and moles.
Sigmodon hispidus primarily is nocturnal but may be active throughout the day. It mostly forages for grasses and seeds but also may feed on insects and eggs of ground-nesting birds. Food gathered during the warmer months is not stored for winter consumption. Rather, fat storage accumulated during spring, summer, and autumn aids in sustaining individuals over winter.
Hispid cotton rats are prolific and produce several litters of 2–10 young, averaging about 5, a year. Their breeding habits vary in response to levels of precipitation. During wet years they breed throughout the year; during periods of drought reproductive activity declines significantly. In the northern part of the state it appears that reproduction is curtailed during the coldest months. Captive females have given birth to as many as nine litters a year; data from wild-caught rats likewise indicate a nearly yearlong breeding season, at least in the warmer parts of their range. The gestation period is approximately 27 days. Females frequently breed again immediately after partus. At birth, the pups are hairless for the most part, pink, and blind and weigh about 5 g. They develop rapidly. The eyes open in about 36 hours, the incisors erupt on the fifth or sixth day, and the young rats are usually weaned when 15 or 20 days old. They can be successfully weaned, however, as soon as the teeth have erupted (5–6 days). Sexual maturity is reached in about 40 days, when the animals are still in juvenile pelage; at 6 months young rats are indistinguishable externally from adults.
Hispid cotton rat populations are cyclical and subject to extreme fluctuations in density, and they have the reproductive capacity to increase rapidly under favorable conditions. Incredible densities of cotton rats have been documented following several successive wet, rainy years and mild winters. These outbreaks have caused serious losses (as much as 90% loss in some instances) to farm crops, particularly peas, peanuts, watermelons, and cauliflower. Population explosions have been documented around San Antonio in 1889, statewide in 1919, in McLennan County in 1928, statewide in the late 1930s and 1940s, throughout eastern Texas from 1958 to 1960, and near Wichita Falls in 1961. Interestingly, extreme outbreaks have not been reported in the past several decades, although a local outbreak was reported in the early 2000s near Brownfield, Texas.
Population explosions are not difficult to comprehend when one is aware of the reproductive potential of these rats. For example, an adult female may breed throughout the year in Texas when conditions are favorable, and she may produce as many as nine litters of 10 young each (normally less). The gestation period is only 4 weeks, and the female breeds again within a few hours after giving birth. Young females are sexually mature in 40 days and can be mothers at the tender age of 68 days and grandmothers at 136 days! Thus, if we assume a new generation of cotton rats every 68 days, a female could be a great-great-great-grandmother at the age of 1 year and be the ancestor of about 15,500 cotton rats. If this rate of reproduction were extended for only three more generations and all survived, the grand total of offspring from the original female would be over 3.5 million!
Although this potential is always present in cotton rats, it is seldom realized because of death due to predators, disease, lack of suitable or sufficient food, accidents, smaller litters, fewer litters a year, and so on. But when conditions are right, the population explodes, and we are knee-deep in cotton rats before we know it.
Fortunately, every eruption is followed by a crash in the population that is brought on by a combination of factors, principally disease. Predators such as coyotes, bobcats, hawks, owls, and certain snakes take their toll, but the main killer is disease. As the rats increase in number, the animals become more and more crowded and provide more contacts for the rapid spread of disease. At the same time, the virulence of the disease increases, until finally the crash occurs and the population is low once again.
POPULATION STATUS. Common. The hispid cotton rat is broadly distributed and probably occurs abundantly in every county of the state. It has fared well as a result of the increase throughout the twentieth century of railroad and highway right-of-ways that provide suitable habitat for dispersal and as a product of the millions of acres of grasslands resulting from the Conservation Reserve Program.
CONSERVATION STATUS. The IUCN lists the hispid cotton rat as a species of least concern, and it does not appear on the federal or state lists of concerned species. It is under no threat.
REMARKS. The hispid cotton rat has been implicated as a possible reservoir for Black Creek Canal virus, a strain of hantavirus responsible for hantavirus pulmonary syndrome. Caution should be exhibited when handling this species.
The taxonomy of S. hispidus has undergone several changes. This species was once considered a single species distributed from the United States to northern South America. In the 2000s, Lottie Peppers, Dallas Henson, and RDB at Texas Tech University separated S. hispidus into three species: S. hirsutus in Central and South America; S. toltecus in Mexico and northern Central America; and S. hispidus in the United States to just south of the Rio Grande. The distributional boundary between S. hispidus and S. toltecus is unclear, although it lies somewhere to the south of the Rio Grande. Additional studies are needed along the Texas–Mexico border to determine if S. toltecus occurs in Texas.
Further, RDB and colleagues at Texas Tech University and Caleb Phillips and Russell Pfau of Tarleton State University independently detected a major genetic division between eastern and western hispid cotton rats. The boundary appears to be along the eastern Piney Woods and Post Oak Savannah near Nacogdoches and then south. Further research is needed to determine if S. hispidus should be recognized as two species.
From The Mammals of Texas, Seventh Edition by David J. Schmidly and Robert D. Bradley, copyright © 1994, 2004, 2016. Courtesy of the University of Texas Press.
Natural Science Research Laboratory
-
Address
Museum of Texas Tech University, 3301 4th street, Lubbock, TX 79409 -
Phone
806.742.2486 -
Email
nsrl.museum@ttu.edu

