
Faculty News
Gill Receives Chancelor's Council Distinguished Research Award
Dr. Harvinder S. Gill's excellence in research was recognized by his receipt of the Chancellor's Council Distinguished Research Award. One of the focuses in Dr. Gill's laboratory has been the development of a painfree microneedle-based therapy for treating allergies. The prevalence of allergic disease has continued to rise worldwide for more than 50 years, with an estimated 40 percent of the population sensitized to produce IgE antibodies to allergens. In the United States along, nearly 6 million children suffer from food allergies against peanuts, milk, eggs, shellfish, and wheat. Allergic reaction is an emerging epidemic of the industrialized world.

Currently, allergen-specific immunotherapy (ASI) is the only disease-modifying treatment available that can desensitize a patient to allergens. Subcutaneous immunotherapy, also known as allergy-shots, are the standard for performing ASI, and this treatment involves repeated injections of the allergen. Conventional needle-based allergy-shots have several drawbacks, including the need for repeated allergen injections over multiple years, the need to be in a clinic to receive the treatment, and a risk of life-threatening systemic anaphylactic reaction, an allergic reaction that can occur quickly – as fast as within a couple of minutes of exposure to the allergen.
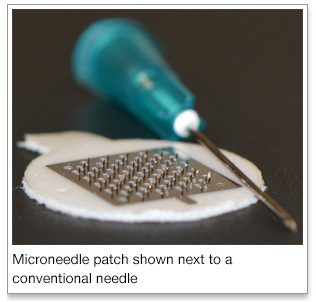
Dr. Gill's work focuses on developing a new microneedle approach to deliver allergen-specific immonotherapy. He has a $1.8 million grant from the National Institutes of Health (NIH) to study a novel minimally-invasive cutaneous immunotherapy (CIT) using allergen-coated microneedles. Microneedles comprise of microscopic projections on a patch that can be self-applied on the skin. Starting out as a thin sheet of stainless steel, the microneedles are cut out of the steel and bent upward, resulting in a patch with a flat surface containing approximately 57 needles. The microneedles can be coated with the chosen allergen through a micro-precision coating device made specifically for the coating process. The allergen is thereby effectively and efficiently applied only to the microneedles themselves and not to the base of the stainless steel patch, minimizing waste of expensive resources, and ensuring dose-consistency. Dosage amounts can also be controlled, with the possibility of being able to use lower doses than necessary with allergy-shots. This method could take the fear, pain, and inconvenience out of allergy immunotherapy. Preliminary data show that microneedle-CIT is at least as effective as allergy-shots. With continued success, Dr. Gill's research will provide the data necessary to support further development of microneedles for painless allergy immunotherapy.

Edward E. Whitacre Jr. College of Engineering
-
Address
100 Engineering Center Box 43103 Lubbock, Texas 79409-3103 -
Phone
806.742.3451 -
Email
webmaster.coe@ttu.edu


