Dr. Adelia Aquino

Title: Adjunct Professor
Education: Ph.D. Chemistry, Universidade de São Paulo, Brazil, 1991
Postdoctoral Study, University of California, San Diego (1994-95) and San Diego Super
Computer Center, 1996
Research Area: Theoretical and Computational Chemistry
Email: adelia.aquino@ttu.edu
Principal Research Interests
- Modeling Basic Interactions in Humic Substances
- Water Molecule Bridges and Cation Bridges Linking Individual Compounds in Soil Organic Matter
- Hydrogen-Bonded Interactions
- Excited-State Proton Transfer
- Charge Transfer and Excitonic Interactions in DNA nucleobases
Environmental pollution is one of the major problems of our civilization mainly due to urban, industrial and agricultural human activities. Produced chemical pollutants represent serious risk for the quality of air, water sources and soils. Dr. Aquino´s research interests aim at providing in-depth information of defined interactions between organic chemicals and soil constituents on molecular level. For this purpose a large variety of quantum chemical methods, mostly based on density functional theory, and of molecular dynamics methods are used to study the properties of humic substances and their interaction with pollutants (e.g. pesticides). Water clusters embedded in the organic soil matrix play a key role in determining the aging effects of soils. Supramolecular models depicted in the following scheme have been used to investigate the stabilizing effects of water molecule bridges (WAMB).
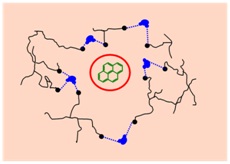
The current work of Dr. Aquino is directed in developing more extended models for nano-pores in humic substance models based on quantum chemical methods. This latter aspect is important since a great variety of interaction types occur which cannot be represented in general by force field methods alone. The quantum chemical tools developed allow the construction and reliable computation of sophisticated and realistic models in soil science and environmental sciences in general. These methods will also be applied to simulations of acid-base properties of surface models for MnO2 polymorphs. Interaction sites of interest on the [110] surface of pyrolusite are illustrated in the figure below.
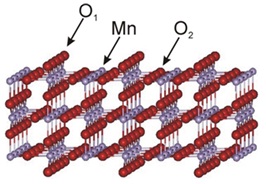
[110] surface of pyrolusite
In collaboration with experimental groups Dr. Aquino has studied intramolecular excited state proton transfer (ESIPT) for the prototype systems 10-hydroxybenzo[h]quinoline (HBQ) and 2-(2'-hydroxyphenyl)benzothiazole (HBT) using coupled cluster and density functional methods. Additionally, she has performed benchmark investigations on the charge transfer in stacked structures of DNA nucleobases through coupled cluster (CC2) calculations. Based on this experience, Dr. Aquino is presently studying on excitonic spectra of stacked dimer and tetramer structures of DNA nucleobases.
Representative Publications
- "Solvent effects on hydrogen bonds - A theoretical study.", Adélia J.A. Aquino; Daniel Tunega; Georg Haberhauer; Martin H. Gerzabek and Hans Lischka J. Phys. Chem. 2002, 106, 1862-1871.
- "Excited-state intramolecular proton transfer: A survey of potential energy surfaces.", Adélia J.A. Aquino; Hans Lischka and Christoph Hättig J. Phys. Chem. A. 2005, 109, 3201-3208.
- "Acid-base properties of a goethite surface model: a theoretical view.", Adélia J.A. Aquino; Daniel Tunega; George Haberhauer; Martin H. Gerzabek and H. Lischka Geochimica et Cosmochimica Acta 2008, 72, 3587-3602.
- "The Functionality of Cation Bridges for Binding Polar Groups in Soil Aggregates.", Adélia J. A. Aquino; Daniel Tunega; Gabriele. E. Schaumann; George Haberhauer; Martin H. Gerzabek and Hans Lischka Int. J. of Quant. Chem. 2011, 111, 1531-1542.
- "The charge-transfer states in a stacked nucleobase dimer complex: a benchmark study.", Adélia J. A. Aquino; Dana. Nachtigallova; Pavel Hobza; Donald G. Truhlar; Christoph Hättig and Hans Lischka J. Comp. Chem. 2011, 32, 1217-1227.
- "Molecular dynamics simulations of water molecule-bridges in polar domains of humic acids.", Adélia J. A. Aquino; Daniel Tunega; Hasan Pasalic; Gabriele E. Schaumann; Georg Haberhauer; Martin Gerzabek and Hans Lischka Environmental Science & Technology 2011, (DOI: 10.1021/es201831g)
Department of Chemistry & Biochemistry
-
Address
1204 Boston Avenue, Lubbock, TX 79409-1061 -
Phone
806.742.3067
