Dr. Gregory I. Gellene

Title: Professor
Education: Ph.D., Cornell University, 1983
Postdoctoral Study, Cornell University, 1984
Research Area: Physical Chemistry
Office: Chemistry 044
Phone: 806-834-8758
Fax: 806-742-1289
Email: greg.gellene@ttu.edu
Webpage: Research Group
Principal Research Interests
- Nuclear Symmetry Effects in Chemical Reactions
- Ab Initio Quantum Chemistry
- Atmospheric Chemistry
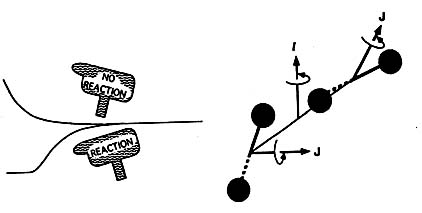
Parallel with the experimental studies, we have been developing a theoretical model of SIKIE which is still ongoing. The complementing roles of theory and experiment in this area has been excellent with theory suggesting new experiments and experiments indicated how the theoretical model should be modified. A recent significant application of the theory provided the first comprehensive explanation for SIKIE in stratospheric ozone; a phenomena which had defied explanation for more than a decade.
A second area of research involves various neutralized ion beam experiments, a novel mass spectrometric approach whereby highly reactive, transient molecules can be produced in a beam mode by the neutralization of an appropriate precursor molecule. Currently, this technique is being used to study radical intermediates in the atmospheric reactions of nitrogen and sulfur containing compounds. Examples include the N2H radical, a long suspected, though never observed, intermediate in the thermal "DeNOx" reaction (an industrial process for lowering NOx smoke stack emissions), and the (CH3)2SOH radical, a key intermediate in the atmospheric reduction of (CH3)2S ultimately leading to SO3 and acid rain.
Computational chemistry constitutes a third general area of on-going research. Because there is little or no experimental information available for many of the chemical species under investigation, valuable insight is provided by is provided by ab initio calculations. Some of the studies such as the development of a global potential energy surface for He3+ are motivated by the experimental part of the program. Other studies, such as work on the polyiodide anions is independent of the experimental program.
Representative Publications
- "Ab Initio Calculations of the Ground State of Polyiodide Anions."Sharp, S. B.; Gellene, G. I. J. Chem. Phys. 1997, 101, 2192.
- "Symmetry Induced Kinetic Isotope Effects in Ion-Molecule Reactions."Gellene, G. I. In Advances in Gas Phase Ion Chemistry Vol. 2, edited by N. G. Adams and L. M. Babcock (JAI, Greenwich, CT, 1996).
- "Symmetry Induced Kinetic Isotope Effects in the Formation of Ar·CO2+."Yoo, R. K.; Gellene, G. I.J. Chem. Phys. 1996, 105, 177.
- "An Explanation for Symmetry Induced Isotopic Fractionation in Ozone."Gellene, G. I. Science 1996, 274, 1344.
- "Ab Initio QCISD(T) Calculations of the Ground Potential Energy Surface of He3+. "Satterwhite, M. F.; Gellene, G. I. J. Phys. Chem. 1995, 99, 13397.
Department of Chemistry & Biochemistry
-
Address
1204 Boston Avenue, Lubbock, TX 79409-1061 -
Phone
806.742.3067
